 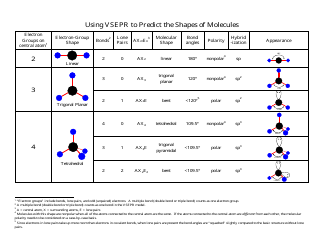
All you need to do is to work out how many. VSEPR theory is used to explain the 3D shape around an atom in a molecule and for small molecules this can give the overall shape. In VSEPR Theory, single bond, double bond and triple bond are all. 
The following table shows the molecular geometries that arise from all. The actual shape depends on the number of bond pairs and lone pairs around the central atom. \): The Difference in the Space Occupied by a Lone Pair of Electrons and by a Bonding PairĪs with SO 2, this composite model of electron distribution and negative electrostatic potential in ammonia shows that a lone pair of electrons occupies a larger region of space around the nitrogen atom than does a bonding pair of electrons that is shared with a hydrogen atom. Figure 7.17 The molecular structure of the methane molecule, CH 4, is shown with a tetrahedral arrangement of the hydrogen atoms. The shape of a molecule or ion is governed by the arrangement of the electron pairs around the central atom. VSEPR theory helps us predict the shapes of simple molecules such as CO2 or. The VSEPR theory is simply a set of rules for predicting the maximum spherical symmetry of electron pairs, which is not the same thing at all. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
